The Atomic Theory, Atomic Structure, and Mass Number vs. Atomic Number
The Atomic Theory states that all matter is made up of tiny little particles
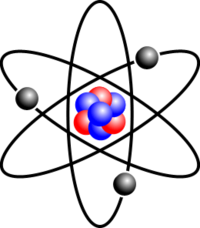
Atoms are made up of protons, neutrons, and electrons. The thing that sets elements apart is the number of electrons, protons, and neutrons. The protons and neutrons are always in the center of the atom. The center part is called the nucleus. If the atom were the size of a stadium the nucleus would be the size of a marble. All atoms have equal numbers of protons and electrons. Electrons have a negative charge. Protons have a positive charge. Neutrons have no charge , they are neutral. Isotopes are atoms of the same element with different mass numbers.

The atomic number is the equal amount of protons while the mass number is the equal amount of protons plus neutrons.
Atomic mass is calculated by figuring out the amount of each type of atom
