- Home
- Topics
- Scientific Method and Laboratory Safety>
- Matter and Its Properties>
- Measurements and Solving Problems>
- The Atom>
- The Electron>
- Periodic Table>
- Ionic Bonding and Naming>
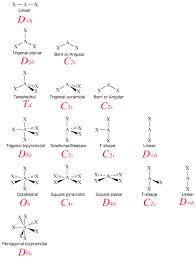
- Molecular Bonding, Naming, and Geometry>
- Chemical Quantities>
- Chemical Reactions>
- Oxidation-Reduction Reactions>
- Solutions>
- Acids, Bases, and Salts
- Behavior of Gases>
- Kinetic Molecular Theory and Thermochemistry
- Bibliography