Bond Forces and Bond Polarity
Polar Bond are bonds where electrons are shared unequally among atoms. Atoms that are more electronegative pull electrons in closer. Atoms that are different will have a different pull on all electrons.
Non-Polar Bonds are bonds where electrons are shared equally among atoms
Polar V. Non-Polar
There or 4 Intermolecular Forces in the following:
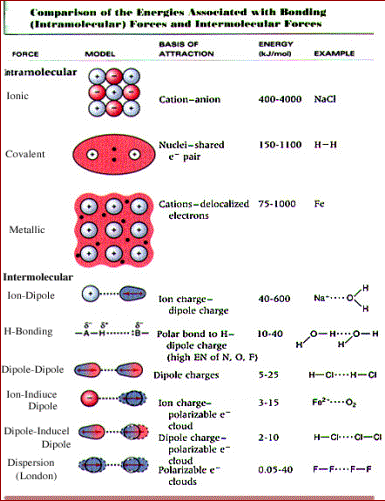
Ionic forces are made up of anions and cations stuck together with electrostatic force
Hydrogen Bonding happens between molecules having an eternal net dipole resulting from Hydrogen being covalently bonded
Dipole-Dipole which happens between molecules that have permanent net dipoles-polar molecules
London Dispersion Forces which temporarily dipoles happening because of uneven electron distribution in surrounding molecules as they approach one another.
Non-Polar Bonds are bonds where electrons are shared equally among atoms
Polar V. Non-Polar
There or 4 Intermolecular Forces in the following:
Ionic forces are made up of anions and cations stuck together with electrostatic force
Hydrogen Bonding happens between molecules having an eternal net dipole resulting from Hydrogen being covalently bonded
Dipole-Dipole which happens between molecules that have permanent net dipoles-polar molecules
London Dispersion Forces which temporarily dipoles happening because of uneven electron distribution in surrounding molecules as they approach one another.