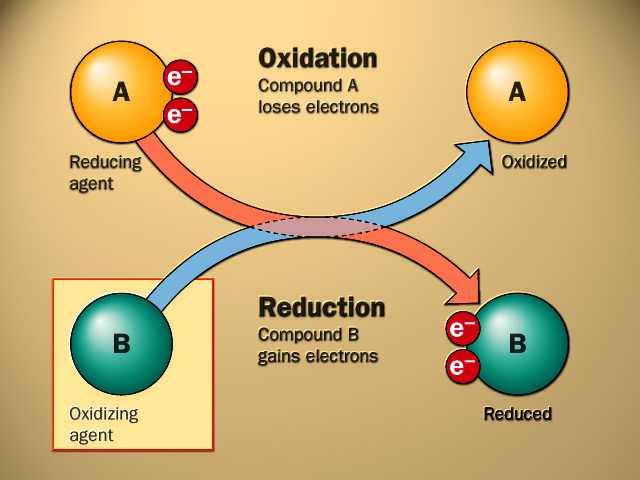
Oxidation/ Reduction Reactions
Oxidation Numbers can be assigned with the help of the periodic table is the compound is an ionic compound. Oxidation numbers are different from formal charge. Oxidation numbers are yet another way to count for electrons during chemical changes.
Rules for Determining Oxidation Numbers:
1. The oxidation number of any element standing alone is 0
2. The oxidation number of a monatomic ion equals its charge.
3. Oxygen’s oxidation number is (-2) UNLESS in peroxides (H2O2) then it is (-1) or when it bonds with Fluorine then it will be (+2)
4. The oxidation number of Hydrogen is (+1) UNLESS it bonds with metals to from metal hydrides then it is (-1) or in the polyatomic ion NH4 where it is also (-1)
5. The sum of the oxidation numbers for a compound must equal zero charge
6. The sum of the oxidation numbers in the formula of a polyatomic ion is equal to its charge
Rules for Determining Oxidation Numbers:
1. The oxidation number of any element standing alone is 0
2. The oxidation number of a monatomic ion equals its charge.
3. Oxygen’s oxidation number is (-2) UNLESS in peroxides (H2O2) then it is (-1) or when it bonds with Fluorine then it will be (+2)
4. The oxidation number of Hydrogen is (+1) UNLESS it bonds with metals to from metal hydrides then it is (-1) or in the polyatomic ion NH4 where it is also (-1)
5. The sum of the oxidation numbers for a compound must equal zero charge
6. The sum of the oxidation numbers in the formula of a polyatomic ion is equal to its charge