Percent Composition, Empirical and Molecular Formulas

A compounds percent composition is the total mass percent.
The steps to finding the Percent composition are easy just follow along!
Step 1: Add up the molar mass of each element that is given (EX. CO2 = 44.01 because carbon is 12.01 and oxygen is 16 and you multiply that by 2 since you are giving 2 oxygen's)
Step 2: You divide the separate molar mass by the total accumulative mass
Step 3: Multiply that number by 100 and round to the number that are in the directions.

To find the Empirical Formula of a compound is are some steps to help you out!
Step 1: you have to find the number of moles of each element
Step 2: Then you divide the number of grams by the atomic weight of the element given
Step 3: Find the ratio between the number of moles and then divide it by the smaller number
Step 4: Make sure you round to the nearest whole number and then the 2 or 3 numbers left that will be your empirical formula
Step 1: you have to find the number of moles of each element
Step 2: Then you divide the number of grams by the atomic weight of the element given
Step 3: Find the ratio between the number of moles and then divide it by the smaller number
Step 4: Make sure you round to the nearest whole number and then the 2 or 3 numbers left that will be your empirical formula
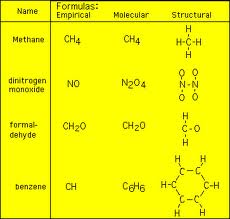
To find the Molecular Formula for a compound here are more steps to follow along too!
Step 1: Since you already found the empirical formula you will now need to find the empirical formula mass. You take the elements you are given and add up its atomic mass
Step 2: you will use the formula mm/efm which mm stands for molar mass and you will be given that in the problem
Step 3: Plug in the numbers and calculate and round to the nearest whole number, you then times that number by your empirical formula and you will end up with the molecular formula
Step 1: Since you already found the empirical formula you will now need to find the empirical formula mass. You take the elements you are given and add up its atomic mass
Step 2: you will use the formula mm/efm which mm stands for molar mass and you will be given that in the problem
Step 3: Plug in the numbers and calculate and round to the nearest whole number, you then times that number by your empirical formula and you will end up with the molecular formula
