Balancing Chemical Equations and 5 Types of Reactions
To balance equations you list the amount of atoms on both sides of the equation. Lets say you have Ag+Cl2 = AgCl3
Your “tally method” would look like this :
1 - Ag - 1
2 - Cl - 3
So you would add coefficients where necessary
2Ag+3Cl2=2AgCl3
so now your “tally method” looks like this
2 - Ag - 2
6 - Cl - 6
Your “tally method” would look like this :
1 - Ag - 1
2 - Cl - 3
So you would add coefficients where necessary
2Ag+3Cl2=2AgCl3
so now your “tally method” looks like this
2 - Ag - 2
6 - Cl - 6
5 Types of Reactions
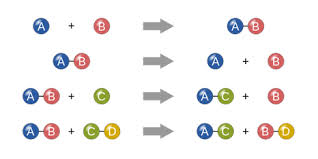
1. Synthesis which is two elements that combine to form a product
2. Decomposition is compounds that breaks down into elements or smaller compounds
*Metal Chlorate Decomposition
*Metal Carbonate Decomposition
*Metal Hydroxide
3. Single Replacement is when an element or ion moves out of one compound and into another
4.Double Replacement is when two compounds react to form two new compounds
*Neutralization Reaction: A reaction between an acid and a base
5.Combustion which is a reaction with oxygen
1. Synthesis which is two elements that combine to form a product
2. Decomposition is compounds that breaks down into elements or smaller compounds
*Metal Chlorate Decomposition
*Metal Carbonate Decomposition
*Metal Hydroxide
3. Single Replacement is when an element or ion moves out of one compound and into another
4.Double Replacement is when two compounds react to form two new compounds
*Neutralization Reaction: A reaction between an acid and a base
5.Combustion which is a reaction with oxygen