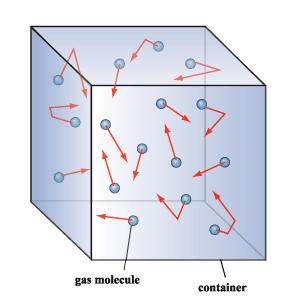
Kinetic Molecular Theory and Thermochemistry
Kinetic Theory says that the tiny particles in matter are always in motion.
*There are 3 principles of Kinetic Theory
-All matter is made up of tiny little particles
-The tiny particles in matter are always in motion
-When the tiny particle collide with each other or the container this simply means the are perfectly elastic and no energy is lost.
There are 5 states of matter including solid, liquid, gas, plasma, and Bose- Einstein Condensate
Solids
Amorphous Solid is a type of solid with no defined shape and it is not a crystal. A solid that lacks an ordered internal structure.
Allotropes Solid is a type of solid that appears in more than one form. Two or more molecular forms of the same elements in the same physical state.
Liquids
Viscosity is resistance of a liquid to flowing
Gases
Plasma
Bose-Einstein Condensate
Gases and Pressure
Temperature
Kinetic Energy and Temperature
Speed of Gases are if gases have the same temperature you can see which particles are moving faster/slower by using mass found on the periodic table
Phase Changes
Heat in Changes of State
Energy Diagrams(Heating Curves) describe the enthalpy
*There are 3 principles of Kinetic Theory
-All matter is made up of tiny little particles
-The tiny particles in matter are always in motion
-When the tiny particle collide with each other or the container this simply means the are perfectly elastic and no energy is lost.
There are 5 states of matter including solid, liquid, gas, plasma, and Bose- Einstein Condensate
Solids
- they are particles that are really close and packed together. The particles do not move a lot. Definite shape and definite volume. Solids can also be known as crystals that make up unit cells which are different repeating patterns.
Amorphous Solid is a type of solid with no defined shape and it is not a crystal. A solid that lacks an ordered internal structure.
Allotropes Solid is a type of solid that appears in more than one form. Two or more molecular forms of the same elements in the same physical state.
Liquids
- Liquids are particles that are spread apart and move slowly. Liquids have a definite volume but no definite shape.
Viscosity is resistance of a liquid to flowing
Gases
- Gas particles are far apart and move really fast. Gases have no definite shape or volume.
Plasma
- Plasma particles are really far apart and move really fast. Plasma only is existent above 3000 degrees Celsius.
Bose-Einstein Condensate
- Are particles that are really close together and barely move at all. You can only find this at really cold temperatures.
Gases and Pressure
- Gas pressure is the force given by gas per unit surface area of something
Temperature
- Temperature is the measure of the average kinetic energy of the particles. The three units of measurements of temperature are Fahrenheit, Celsius, and kelvin.
Kinetic Energy and Temperature
- Energy of motion, moving object. Kinetic energy is measured in Joules(J)
Speed of Gases are if gases have the same temperature you can see which particles are moving faster/slower by using mass found on the periodic table
Phase Changes
- melting: solid to a liquid
- Freezing: liquid to a solid
- Melting and freezing happen at the same temperature
- Liquefaction: gas toa liquid
- boiling: liquid to a gas/vapor
- sublimation: solid to a vapor
- Deposition: vapor to a solid
Heat in Changes of State
Energy Diagrams(Heating Curves) describe the enthalpy