Dalton's Law of Partial Pressure
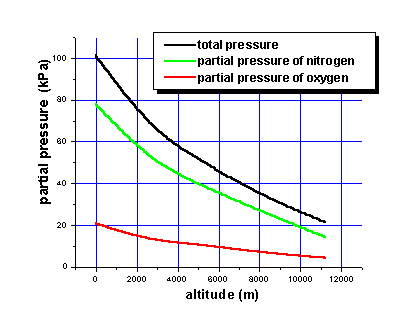
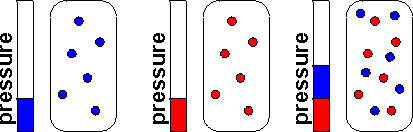
Dalton's Law of Partial Pressure is the total pressure of a mixture of gases that equal the sum of the partial pressures of the individual gases.
* The formula for Partial Pressure is Ptotal = P1 + P2 + ...., depending on how many individual gases you have will depend on the total.
Like always remember that your units need to match up!
Ex. Hydrogen gas is collected over water at 22.5 degree Celsius. Find the pressure of the dry gas if the atmospheric pressure is 94.4 kPa.
Ptotal = 94.4 kPa 94.4 = x = 2.72
PH2O = 2.72 kPa x = 91.7 kPa
PH2 = ? * Watch sig figs!
* The formula for Partial Pressure is Ptotal = P1 + P2 + ...., depending on how many individual gases you have will depend on the total.
Like always remember that your units need to match up!
Ex. Hydrogen gas is collected over water at 22.5 degree Celsius. Find the pressure of the dry gas if the atmospheric pressure is 94.4 kPa.
Ptotal = 94.4 kPa 94.4 = x = 2.72
PH2O = 2.72 kPa x = 91.7 kPa
PH2 = ? * Watch sig figs!