Atoms, Elements, Compounds, Mixtures
The atom or molecule in matter is constantly moving. The lower the temperature, the slower they can move and the higher the temperature, the faster they can move.
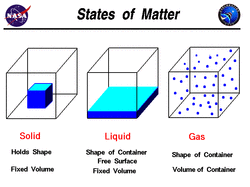
There are 3 states of matter which are:
1.Solid: Atoms are closely placed together in a specific order (Definite Shape, Definite Volume, on-Compressible, High Density)
2.Liquid: Atoms are close but not in order. They have constant volume and form in to shapes depending on placement.(Indefinite shape, Definite volume, Non-Compressible , Less dense than solids)
3.Gas: Atoms are separated and have no fixed shape or volume. (Indefinite shapes, Indefinite Volumes, Compressible,Very low density)
1.Solid: Atoms are closely placed together in a specific order (Definite Shape, Definite Volume, on-Compressible, High Density)
2.Liquid: Atoms are close but not in order. They have constant volume and form in to shapes depending on placement.(Indefinite shape, Definite volume, Non-Compressible , Less dense than solids)
3.Gas: Atoms are separated and have no fixed shape or volume. (Indefinite shapes, Indefinite Volumes, Compressible,Very low density)
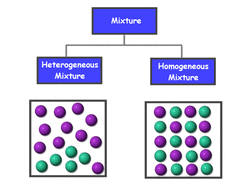
Mixtures are two or more substances that are not chemically combined
There are two types of mixtures which are heterogeneous and homogeneous mixtures
Heterogeneous mixtures are mixtures that do not have a uniform composition. You can pick out parts of the mixture. Some examples would be cereal or chicken
Homogeneous mixtures are mixtures that have a uniform composition which you cannot pick out parts of the mixture such as sugar or water
There are two types of mixtures which are heterogeneous and homogeneous mixtures
Heterogeneous mixtures are mixtures that do not have a uniform composition. You can pick out parts of the mixture. Some examples would be cereal or chicken
Homogeneous mixtures are mixtures that have a uniform composition which you cannot pick out parts of the mixture such as sugar or water
