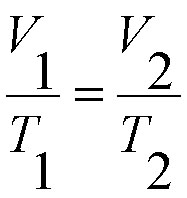Charles' Law
Charles' Law is when the volume and temperature varies at constant pressure, which means when the pressure is constant then the temperature and volume will be related. The equation for Charles' Law is V1/T1=V2/T2. Just as in Boyle's Law, plug in the numbers given in your problem and solve for what is not their.
Since you are dealing with temperature in Charles' Law you will have to convert from Celsius to Kelvin by using the formula K=273 + C. No matter what, you will always have to convert to Celsius.
Also remember, make sure both of your pressures are the same (kPa, atm, mmHg,etc.) and also that your volume is the same, either ml or L.
Ex. The volume in a sample of gas would be 145.5L at 20 degree Celsius. If temperature iis increased to 100 degree Celsius, what will the new volume be?
V1- 145.5L 145.5 = ? 293x =54,271.5 T1- 273 + 20 = 293 Kelvin 293 = 373 293 = 293
V1- ? X = 185.2L
T2- 273 + 100 = 373 Kelvin
Since you are dealing with temperature in Charles' Law you will have to convert from Celsius to Kelvin by using the formula K=273 + C. No matter what, you will always have to convert to Celsius.
Also remember, make sure both of your pressures are the same (kPa, atm, mmHg,etc.) and also that your volume is the same, either ml or L.
Ex. The volume in a sample of gas would be 145.5L at 20 degree Celsius. If temperature iis increased to 100 degree Celsius, what will the new volume be?
V1- 145.5L 145.5 = ? 293x =54,271.5 T1- 273 + 20 = 293 Kelvin 293 = 373 293 = 293
V1- ? X = 185.2L
T2- 273 + 100 = 373 Kelvin