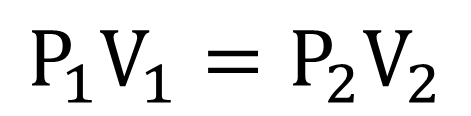Boyle's Gas Law
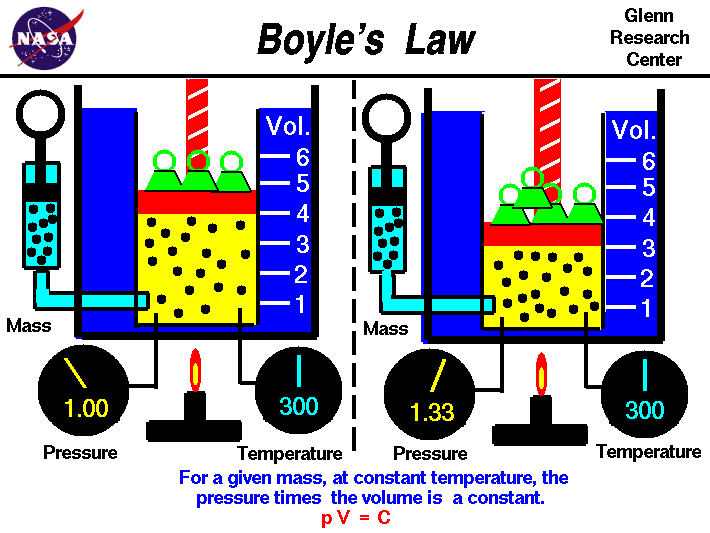
Boyle's Law causes an inverse relationship which means the pressure and volume vary at a constant temperature. The formula for Boyle's gas law is P1(V1)=P2(V2), P1 and V1 are the initial conditions while P2 and V2 are the new conditions or final conditions.
To solve Boyle's Law, first you have to make sure that your units match (kPa, atm, etc) if they don't, you will have to convert. Plug in the numbers you are given and solve for your question mark. Depending on your teacher, watch your sig figs!
Ex. Using 15.3L as the initial volume of oxygen, calculate the volume that would result in the pressure rising from 160 kPa to 260 kPa.
P1- 160 kPa 160x = 260(15.3)
V1- ? 160x = 3,978
P2 - 260 kPa 160 = 160
V2 - 15.3L X = 24.9L
To solve Boyle's Law, first you have to make sure that your units match (kPa, atm, etc) if they don't, you will have to convert. Plug in the numbers you are given and solve for your question mark. Depending on your teacher, watch your sig figs!
Ex. Using 15.3L as the initial volume of oxygen, calculate the volume that would result in the pressure rising from 160 kPa to 260 kPa.
P1- 160 kPa 160x = 260(15.3)
V1- ? 160x = 3,978
P2 - 260 kPa 160 = 160
V2 - 15.3L X = 24.9L