Electron Configurations and Atomic Orbital Diagrams
Electron configuration of an atom shows the arrangement of electrons that are spread out among the orbital shells. The electron configuration is used to describe the orbitals of an atom in its ground state or an atom that has been ionized. The valence electrons become the determining factor for the unique chemistry of the element and what sets it apart. Valence electrons are electrons in the outer most shell.
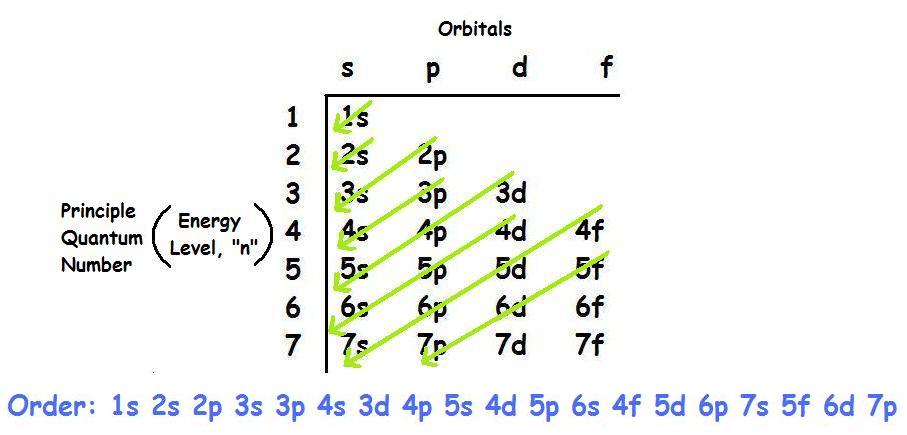
To write the electron configuration, you just count the number of electrons you have and use as many orbital sublevels as you need to store all your electrons. Always fill the lowest energy levels first.
To write the electron configuration, you just count the number of electrons you have and use as many orbital sublevels as you need to store all your electrons. Always fill the lowest energy levels first.

The atomic orbital diagram is a block that the atom is in and where we will count to get the number of electrons in the last subshell.
If you know how to do electron configurations this is easy and you have it down but don’t forget your arrows because they can not point the same direction when right next to each other.
If you know how to do electron configurations this is easy and you have it down but don’t forget your arrows because they can not point the same direction when right next to each other.