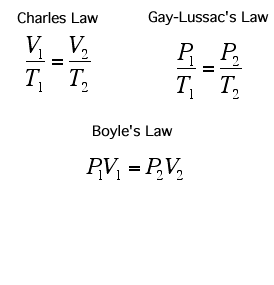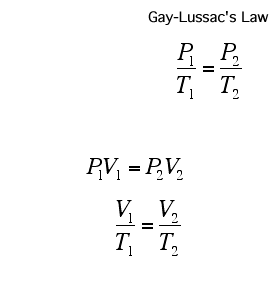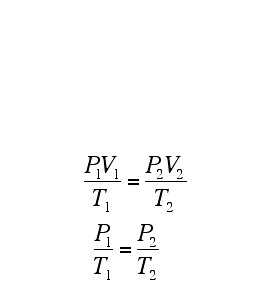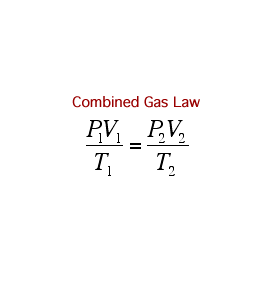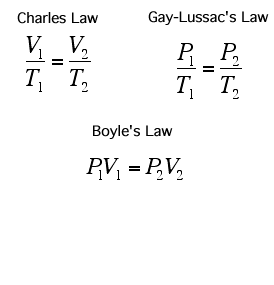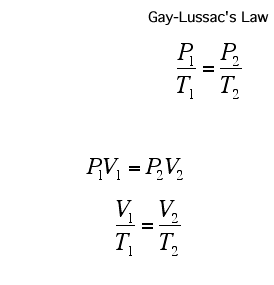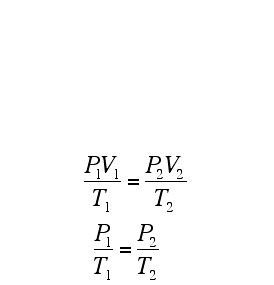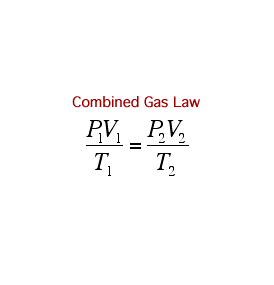What is a Gas Law?
A Gas Law is a relationship between the pressure, volume, temperature, and moles of a gas and using a mathematical equation to solve. There are 6 different Gas Laws that will be discussed in the attaches subtopics. They are Boyle's Law, Charles' Law, Gay-Lussac's Law, Combined Gas Law, Partial Pressures Law, and Ideal Gas Law.
Gas Laws uses the Kelvin temperature scale because Celsius has a negative value and Kelvin does not.
* To convert a Celsius temperature into Kelvin temperature just ass 273, its that simple! But remember if you are finding the temperature in a problem, you will have to convert back to Celsius...do NOT give your answer in Kelvin.
Standard temperature and pressure or (STP) are 273K and 1 atm, you will need to know these because it will probably come up in a problem.
* Remember that the units of volume will either be milliliter (ml), liter (L), or cubic centimeter (cm2)
Gas Laws uses the Kelvin temperature scale because Celsius has a negative value and Kelvin does not.
* To convert a Celsius temperature into Kelvin temperature just ass 273, its that simple! But remember if you are finding the temperature in a problem, you will have to convert back to Celsius...do NOT give your answer in Kelvin.
Standard temperature and pressure or (STP) are 273K and 1 atm, you will need to know these because it will probably come up in a problem.
* Remember that the units of volume will either be milliliter (ml), liter (L), or cubic centimeter (cm2)