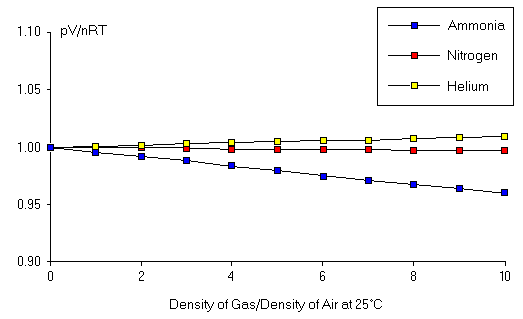
Ideal Gas Law
Ideal Gas Laws are the relationship of pressure, volume, temperature, and moles. Yes, there is moles in this Gas law but don't worry its not that hard! Remember, mole are related to grams and molecules but in the Ideal Gas Law, it must be expressed in MOLES.
The formula for Ideal Gas Law is PV=nRT, which P is for pressure, V is for volume, N is moles, T is temperature, and R is the Ideal Gas Law constant. What that means is the value never changes. ONLY and ONLY in the Ideal Gas Law your unites must match up with the units in the R Value, P must be in atm, V must be in L, T must be in K, and N must be in moles. If your units do not match up, you will have to convert!
The R values: 0.0812 L atm / mole K
62.4 L mmHg / mole K
8.314 L kPa / mole K
Ex. If 234.68 grams of chlorine was compressed at 3534 mmHg of pressure and -13.8 degree Celsius, what volume would it have?
* You will have to convert twice in this one!
P = 4.65 atm 3534mmHg x 1 atm = 4.56 atm 4.65x=3.31(62.4)(259.2)
V = ? 760 mmHg 4.65x=53,536.2
n = 3.31 mol 4.65 = 4.65
T = 273 + (-13.8) = 259.2 K 234.68g x 1 mole = 3.31 moles
R = 63.4 70.9g X = 11, 500 L
The formula for Ideal Gas Law is PV=nRT, which P is for pressure, V is for volume, N is moles, T is temperature, and R is the Ideal Gas Law constant. What that means is the value never changes. ONLY and ONLY in the Ideal Gas Law your unites must match up with the units in the R Value, P must be in atm, V must be in L, T must be in K, and N must be in moles. If your units do not match up, you will have to convert!
The R values: 0.0812 L atm / mole K
62.4 L mmHg / mole K
8.314 L kPa / mole K
Ex. If 234.68 grams of chlorine was compressed at 3534 mmHg of pressure and -13.8 degree Celsius, what volume would it have?
* You will have to convert twice in this one!
P = 4.65 atm 3534mmHg x 1 atm = 4.56 atm 4.65x=3.31(62.4)(259.2)
V = ? 760 mmHg 4.65x=53,536.2
n = 3.31 mol 4.65 = 4.65
T = 273 + (-13.8) = 259.2 K 234.68g x 1 mole = 3.31 moles
R = 63.4 70.9g X = 11, 500 L